
High-Resolution Crystal Structure of an Engineered Human β2-Adrenergic G Protein–Coupled Receptor | Science

The molecular basis of subtype selectivity of human kinin G-protein-coupled receptors | Nature Chemical Biology
Molecular Mechanism of Action for Allosteric Modulators and Agonists in CC-chemokine Receptor 5 (CCR5)*

PDF) Structure-Activity Investigations and Optimisations of Non-metabolite Agonists for the Succinate Receptor 1
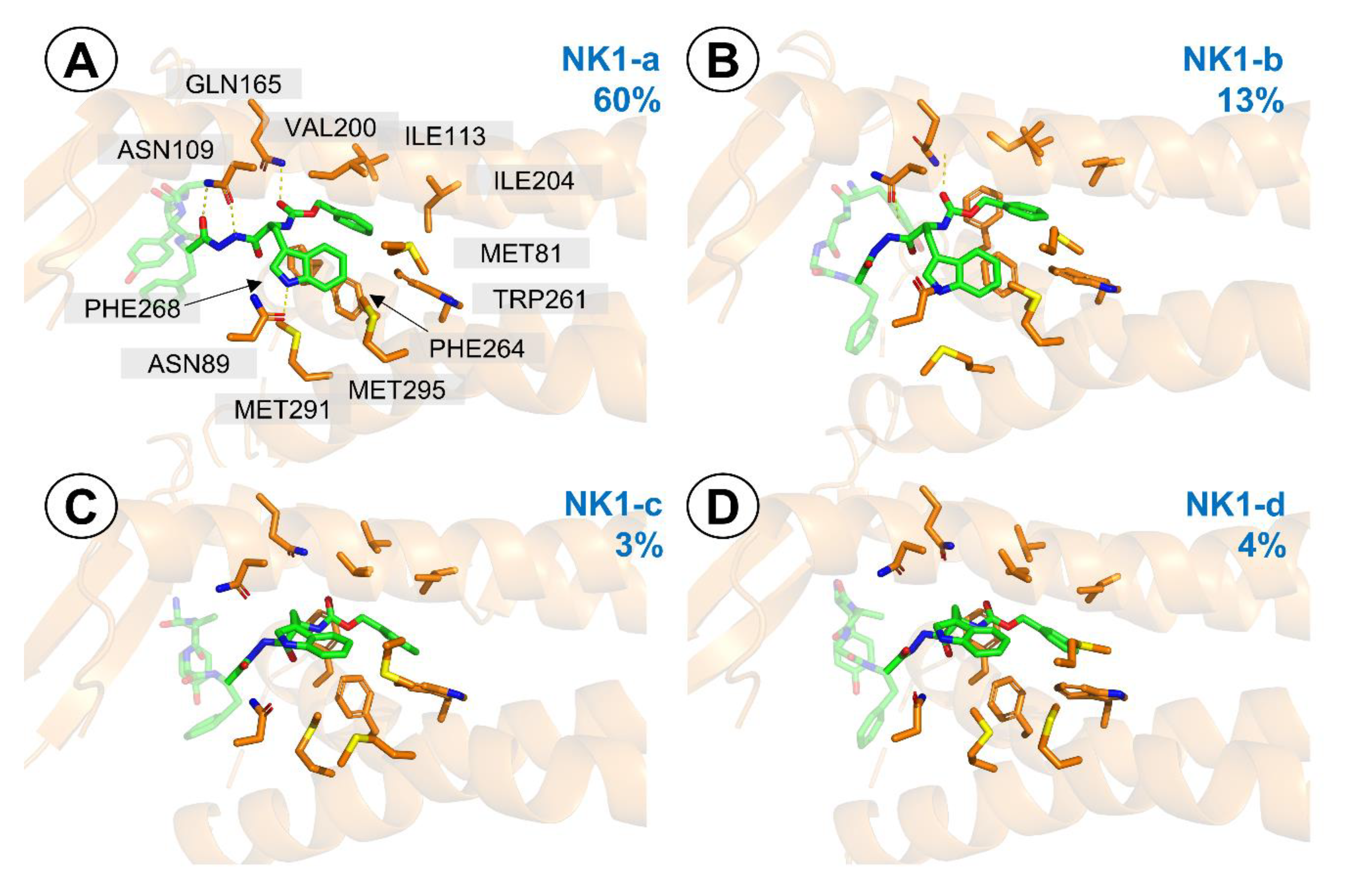
IJMS | Free Full-Text | In Vivo, In Vitro and In Silico Studies of the Hybrid Compound AA3266, an Opioid Agonist/NK1R Antagonist with Selective Cytotoxicity
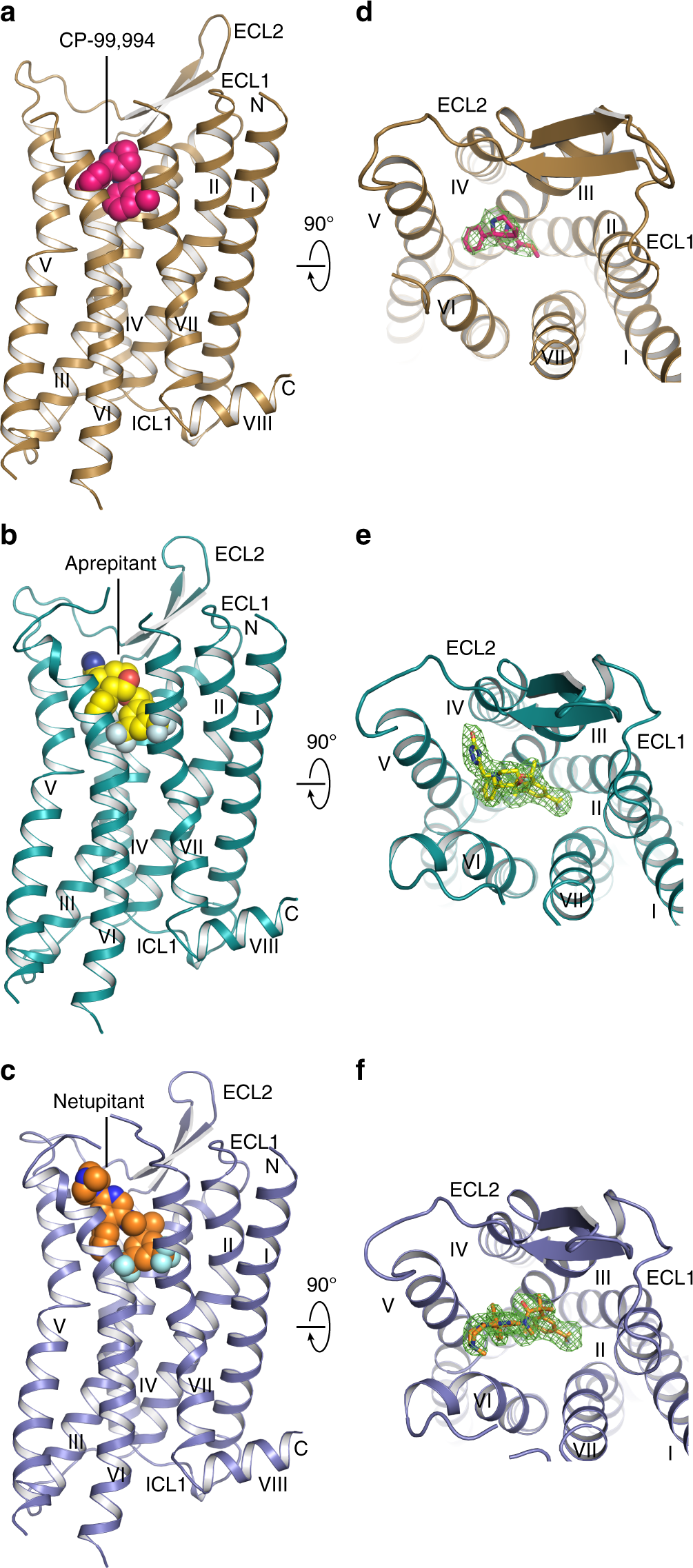
Crystal structures of the human neurokinin 1 receptor in complex with clinically used antagonists | Nature Communications

Synthesis and Biological Evaluation of Biphenyl Amides That Modulate the US28 Receptor - Kralj - 2014 - ChemMedChem - Wiley Online Library
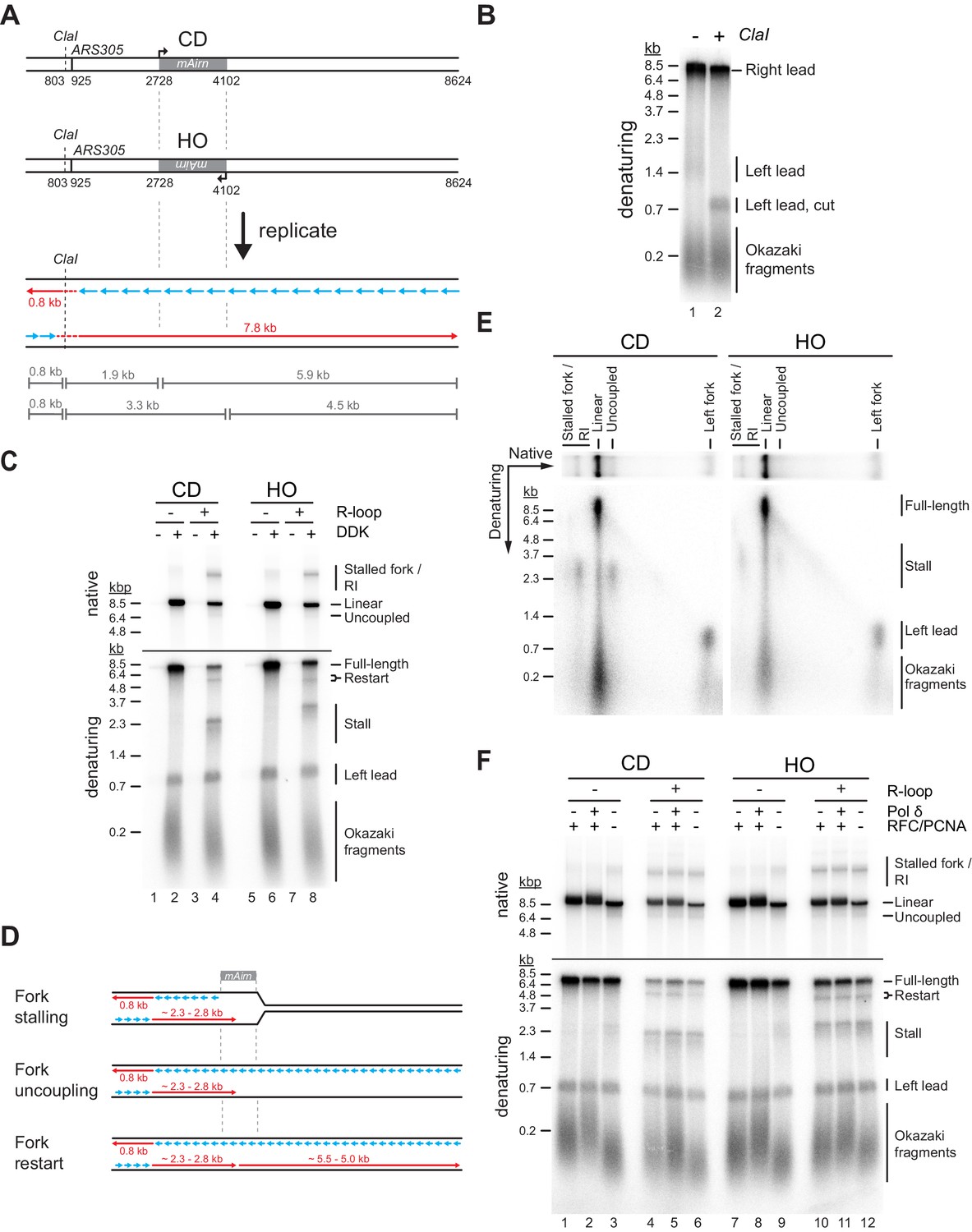
The interplay of RNA:DNA hybrid structure and G-quadruplexes determines the outcome of R-loop-replisome collisions | eLife
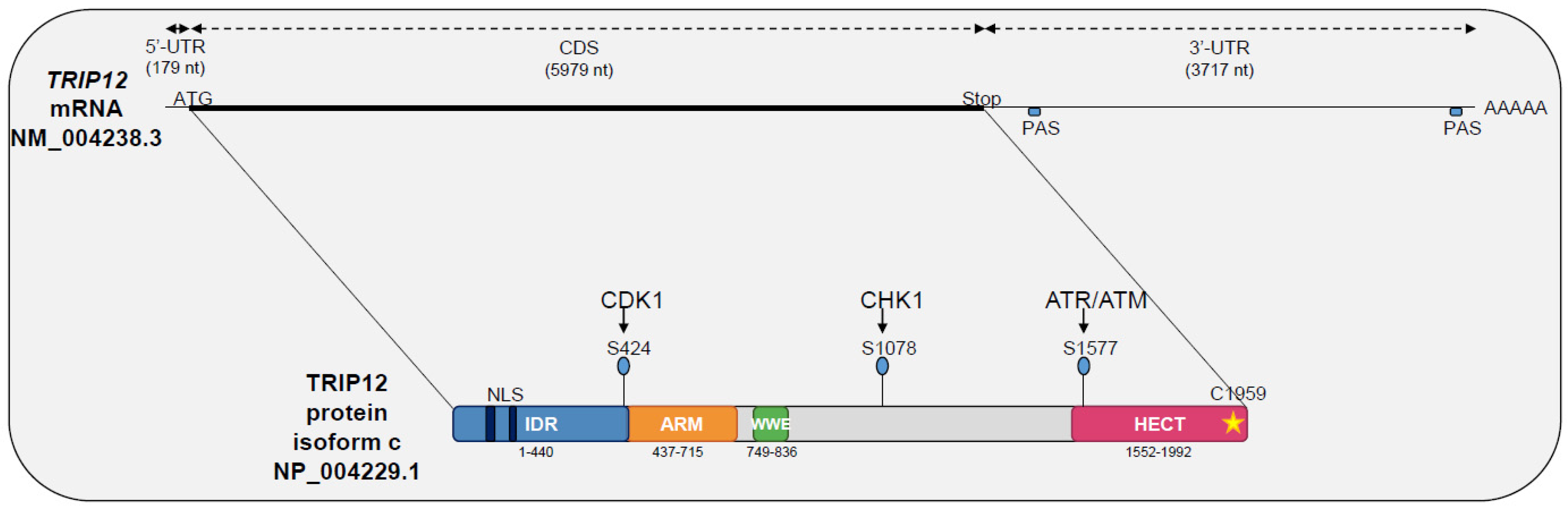
IJMS | Free Full-Text | E3 Ubiquitin Ligase TRIP12: Regulation, Structure, and Physiopathological Functions
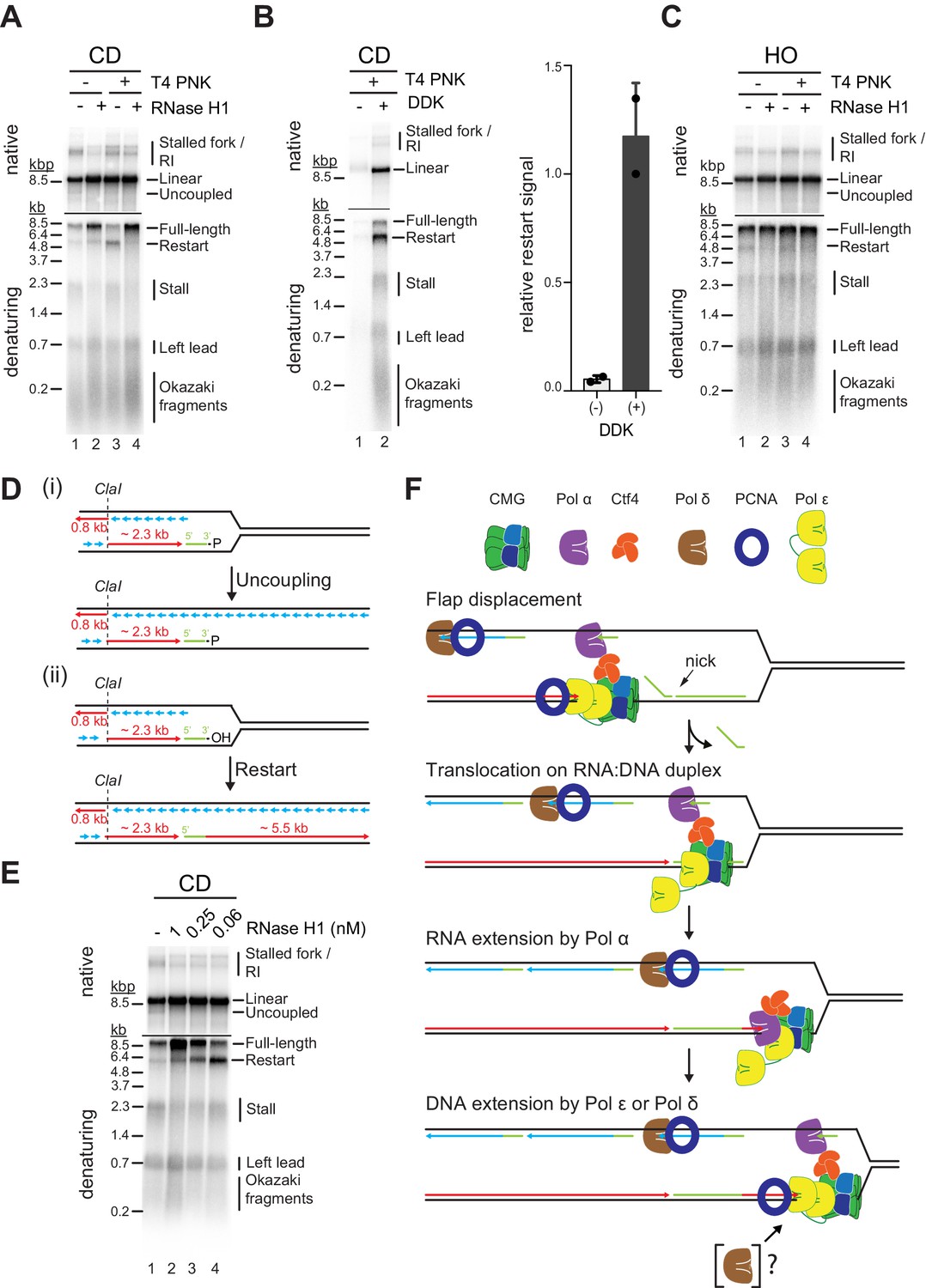
The interplay of RNA:DNA hybrid structure and G-quadruplexes determines the outcome of R-loop-replisome collisions | eLife















